



New SHIC research shows ASF remains infectious for much longer
Pork Checkoff reports that the new recommendation is to hold conventional soybean meal an average of 125 days from when it is “born” or packaged, up from only 52 days determined in previous research.The researchers and veterinarians who created the fact sheet, Holding Time Calculations for Feed Ingredients to Mitigate Virus Transmission, have revised the necessary holding time upward when it comes to determining if the African swine fever (ASF) virus is sufficiently degraded in feedstuffs to potentially prevent transmission.
The new research, Half-Life of African Swine Fever Virus in Shipped Feed, was funded by the Swine Health Information Center with Pork Checkoff funds. The researchers conducted the new study at Kansas State University using actual ASF virus inside the state-of-the-art National Bio and Agro-defense Facility. In a previous study, researchers used Seneca valley virus A (SVA) as a surrogate to ASF virus due to its known longevity of infectiousness and the difficulty in doing research with it.
Initial estimates of feedstuffs holding times in 2018 came from surrogate virus research funded by The Institute for Feed Education and Research, which is the public charity of the American Feed Industry Association. Revisions made in May 2019 and now in February reflect the newest information from work done on the true ASF half-life. Based on the conditions of transoceanic shipment from Asia, the following table displays the mean holding times calculated to provide 99.99 percent ASF degradation at 54 degrees F (average).
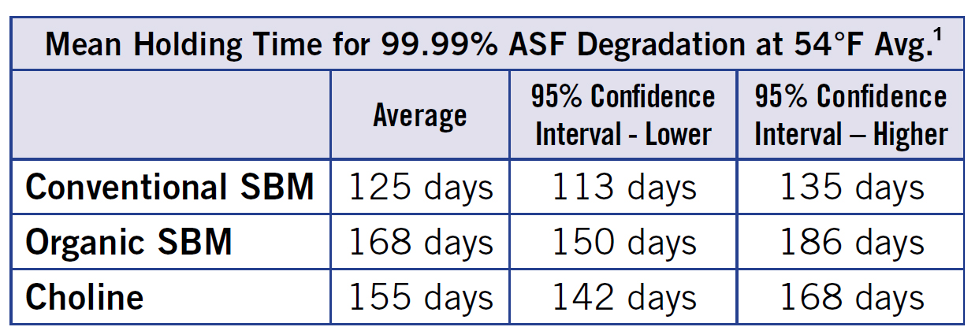
© Stoian, A; Zimmerman, J et al., 2019
Transit time to the United States of potentially contaminated feed ingredients can be applied to the total holding time after the “born-on date” if the ingredient is transported in such a way that would prevent further contamination.
Experts recommend that you talk with your feed suppliers and ask for the born-on date for all imported feed products. Vigilance is the best prevention, so it’s good to review key tactics found in The Role of Feed and Ingredient Biosecurity for ASF Prevention on the pork.org/fad page.
To learn more about the original study on viral half-life, see Stability of SVA in animal feed ingredients at www.swinehealth.org/originalfeedholdingtime on the Swine Health Information Center website.
Also, find biosecurity guidelines in Developing Biosecurity Practices for Feed & Ingredient Manufacturing at www.swinehealth.org/AFIAbiosecuritypractices from the American Feed Ingredient Association website.
| References | ||||
|---|---|---|---|---|
| Stoian, A., Zimmerman, J., et al. | ||||
| (2019) | Half-Life of African Swine Fever Virus in Shipped Feed.. Emerging Infectious Diseases. | 25(12):2261-2263 |






